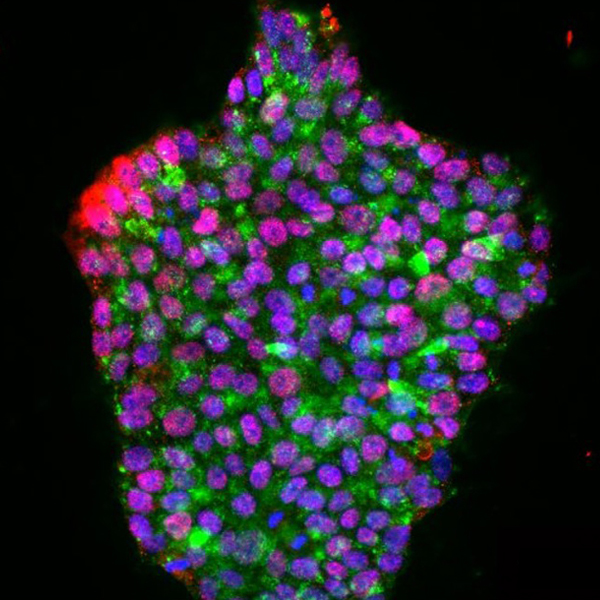
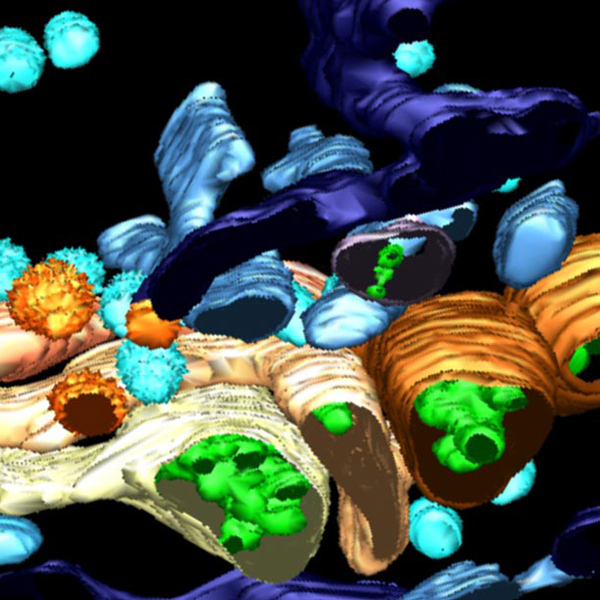
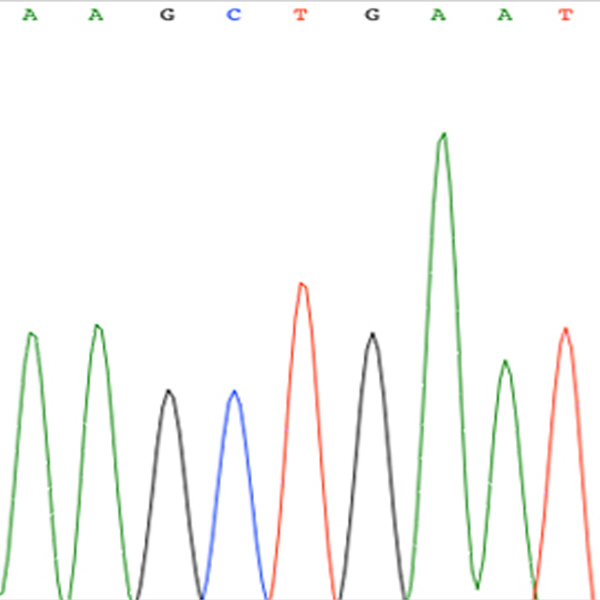
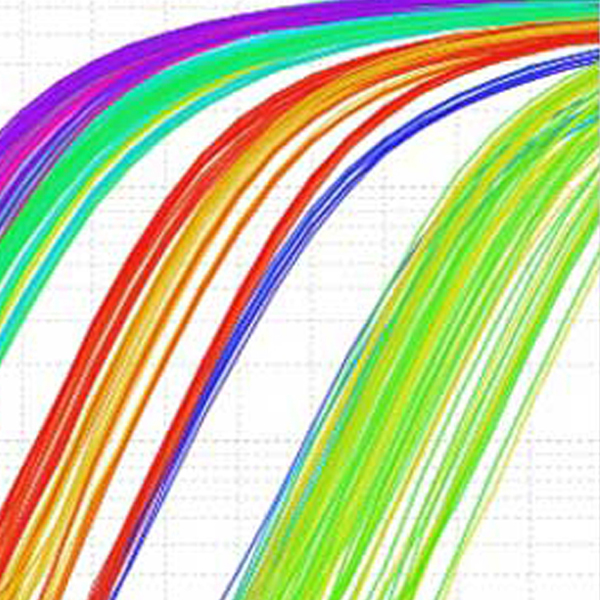
Technological units
Core Facilities: The Technological Heart of Research at IFOM
The Core Facilities at IFOM are central to its scientific progress and innovation. Equipped with cutting-edge instrumentation and led by experts with specialized know-how, these units provide a unique research environment where efficiency, standardization, and innovation converge to accelerate scientific discovery.
Through an integrated and collaborative approach, the Core Facilities go beyond offering technical support—they actively contribute to the co-development of new methodologies and the advancement of research and development. By enabling shared access to highly advanced technological infrastructures, they ensure cost efficiency and provide services that would otherwise be inaccessible to individual research groups.
IFOM Core Facilities offer a wide range of services, including experimental design, data acquisition and analysis, advanced training, and tailored consulting, ensuring optimal access to cutting-edge technologies. These facilities are not merely tools that support research but strategic partners for IFOM scientists, helping transform innovative ideas into tangible results and keeping IFOM at the forefront of cancer research.
The IFOM Core Facilities are complemented by Cogentech's Core Facilities, which primarily support IFOM researchers while also being accessible to external scientists. Cogentech, a Benefit Corporation fully owned by IFOM, is dedicated to delivering comprehensive and reliable scientific and diagnostic services to researchers and clinicians alike.
Training and Access to Core Facilities
The Core Facilities at IFOM provide continuous training to researchers through tailored programs or formalized courses. This approach ensures that every user develops the necessary skills to operate the available technologies independently and safely, while upholding the highest scientific and regulatory standards. Consequently, autonomous access to Core Facility instrumentation is generally granted only after completing specific training, which combines theoretical and practical components.
For the Cellular and Preclinical Models Facility, specialized training is mandatory, particularly for access to Biosafety Level-2 areas. This training emphasizes safety protocols and operational guidelines.
The Cogentech Biological Research Facility, certified ISO 9001 for its training programs, delivers specialized courses on facility access, regulatory compliance, and animal welfare, ensuring adherence to the highest ethical and scientific standards.
This commitment to training not only enhances research excellence but also cultivates a safe and highly skilled working environment.
Acknowledgment and Authorship
According to our Core Facilities Guidelines, if Facility services were used in work described in a publication, the Facility(ies) must be acknowledged, citing the name and Research Resource Identifier (RRID).
We acknowledge the support of IFOM ETS’s
- Advanced Light Microscopy Core Facility RRID: SCR_026866
- Cancer Engineering R&D Core Facility RRID: SCR_026862
- Cellular and Preclinical Models Core Facility RRID: SCR_026864
- Electron Microscopy Cells & Tissues Core Facility RRID: SCR_026863
- Electron Microscopy Single Molecules Core Facility RRID: SCR_026864
- Flow Cytometry Core Facility RRID: SCR_026865
- Functional Genomic Screening Core Facility RRID: SCR_026867
We acknowledge the support of Cogentech SB Srl’s (held by IFOM ETS)
- Biological Research Facility RRID: SCR_026936
- Cancer Genetic Testing Laboratory RRID: SCR_026940 (includes DNA Sanger Sequencing and QPCR)
- Genomics Core Facility RRID: SCR_026935
- Integrative Genomics Core Facility RRID: SCR_026938
- Histopathology Core Facility RRID: SCR_026934
- Proteomics & Metabolomics Core Facility RRID: SCR_026937
In addition, if a staff member of an IFOM or Cogentech Core Facility contributes substantially to a published work, the staff member should be included as an author in the published work. More information on this topic can be found here: Recommended guidelines and Acknowledging and citing core facilities