Cancer Engineering R&D

Overview
Cancer is not only a genetic disease but also influenced by physical forces and the mechanical properties of cells and tissues. Understanding how these forces impact tumor growth, metastasis, and drug resistance has opened new avenues for cancer research. The Cancer Engineering R&D Core Facility provides researchers with cutting-edge microfabrication and 3D printing technologies, allowing the development of experimental models and diagnostic tools that bridge fundamental research and clinical applications.
The Cancer Engineering R&D Core Facility at IFOM develops innovative biomedical technologies and devices to advance cancer research and biomedical applications. By integrating expertise from biology, physics, engineering, and material science, the facility creates state-of-the-art tools to study the mechanical properties of cancer cells, tumor progression, and therapeutic responses.
The Cancer Engineering R&D Core Facility supports a wide range of research projects, focusing on the development and application of advanced biomedical technologies. Key areas include:
- Development of microfluidic chips to model tumor growth, angiogenesis, and metastasis.
- Isolation and detection of circulating tumor cells (CTCs) from patient blood for diagnostic applications.
- Use of Atomic Force Microscopy (AFM) and other technologies to measure mechanical properties of cancer cells, with potential as biomarkers for early cancer detection.
- Engineering of cell stretching and compression devices to study how mechanical forces influence cancer progression.
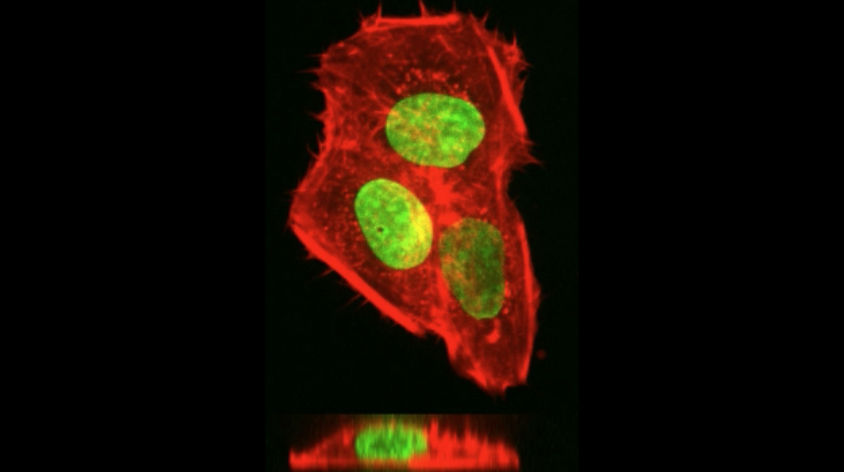
- Development of 3D in vitro tumor models that replicate the cancer microenvironment for drug screening.
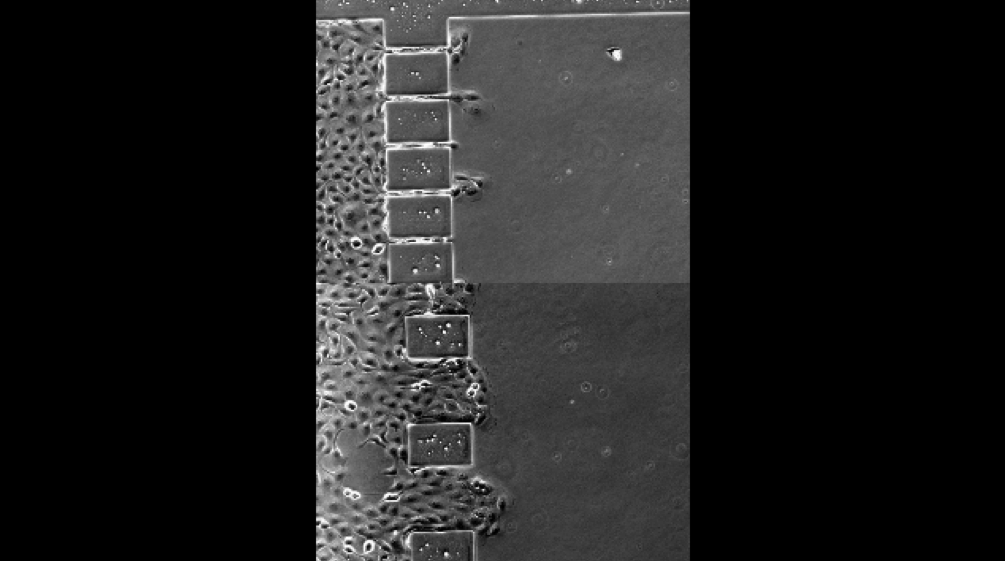
- Fabrication of custom micro-patterned surfaces for studying single-cell and collective cell migration.
The Cancer Engineering R&D Core Facility collaborates with leading IFOM research groups, applying state-of-the-art engineering solutions to fundamental and translational cancer research.
The Cancer Engineering R&D Core Facility is equipped with advanced technologies for microfabrication, 3D printing, cell biomechanics, and the preparation of microstructured devices and surfaces. Below is a list of the main equipment available:
3D Printing and Post-Processing
- Formlabs Form 3 – Industrial-quality SLA resin 3D printer, ideal for high-resolution prototyping
- Formlabs Form 2 – SLA 3D printer for precise, high-detail prints on small-scale structures
- Ultimaker (Makerbot) – High-quality FDM 3D printer for fast prototyping with thermoplastic materials details
- WASP Delta 2040 – Professional-grade 3D printer suitable for technical materials and large-volume fabrication
- Formlabs Wash Station – Automated washing system for post-processing SLA-printed resin models
- WASP Delta 2040 – Professional-grade 3D printer suitable for technical materials and large-volume fabrication
- Formlabs Wash Station – Automated washing system for post-processing SLA-printed resin models
Microscopy and Biomechanics
- JPK NanoWizard 3 (Bruker) – High-sensitivity Atomic Force Microscope (AFM) for single-cell mechanical analysis
Microfabrication and Surface Treatment
- KLOE UV-KUB 3 - Cold UV-LED mask aligner for photolithography on light-sensitive materials, ideal for microstructure fabrication
- Gamma High Voltage - HV Power Electrospinning Machine - High-voltage system for electrospinning nanofibers and scaffold structures
- Harrick PDC-32G-2 - Plasma cleaner for precise substrate cleaning and surface modification details
- Laurell WS-650Mz - Spin coater for uniform thin-film deposition via centrifugal force, commonly used in photolithography
- Jelight UVO-Cleaner Model 342 – UV surface treatment system for cleaning and activation via ultraviolet oxidation
- Valtaro Motori MM71A4 – High-performance dry vacuum pump used in material treatment and microfabrication processes
Cell stretching dish, European Patent application n. PCT/EP2018/053477, 2017
Cell compression device, European Patent application n. EP172000564, 2018
Facility Acknowledgment
According to our Core Facilities Guidelines, if Facility services were used in work described in a publication, the Facility must be acknowledged, citing both the Facillity name and its Research Resource Identifier (RRID). A typically acknowledgement might look like:
The research described was supported by the IFOM Cancer Engineering R&D Core Facility (RRID: SCR_026862)
Co‑Authorship
If a staff member of an IFOM or Cogentech Core Facility contributes substantially to a published work, they should be included as a co‑author.
More information is available in Recommended guidelines and Acknowledging and citing core facilities
Examples of substantial contributions that may justify co‑authorship include:
- Contribution to experimental design or optimization
- Development or significant adaptation of methods or protocols
- Advanced data analysis and interpretation
- Intellectual input influencing the scientific direction of the study
Routine technical support or standard service provision generally warrants acknowledgment rather than co‑authorship.
- C. Malinverno, S. Corallino, F. Giavazzi, M. Bergert, Q. Li, M. Leoni, A. Disanza, E. Frittoli, A. Oldani, E. Martini, T. Lendenmann, G. Deflorian, G. V. Beznoussenko, D. Poulikakos, K. H. Ong, M. Uroz, X. Trepat, D. Parazzoli, P. Maiuri, W. Yu, A. Ferrari, R. Cerbino, G. Scita, 2017
Endocytic reawakening of motility in jammed epithelia
Nature Material, v. 16, p. 587-596. - Q. Li, E. Makhija, F. M. Hameed, and G. V. Shivashankar, 2015
Micropillar displacements by cell traction forces are mechanically correlated with nuclear dynamics
Biochemical and Biophysical Research Communications, v. 461, p. 372-377. - X. Shao*, Q. Li*, A. Mogilner, A. D. Bershadsky, and G. V. Shivashankar, 2015
Mechanical stimulation induces formin-dependent assembly of a perinuclear actin rim
Proceedings of the National Academy of Sciences of the USA, v. 112, p. E2595-E2601. - Q. Li, A. Kumar, E. Makhija, and G. V. Shivashankar, 2014
The regulation of dynamic mechanical coupling between actin cytoskeleton and nucleus by matrix geometry
Biomaterials, v. 35, p. 961-969. - F. Y. Leong, Q. Li, C.T. Lim, and K. H. Chiam, 2011
Modeling cell entry into a micro-channel
Biomechanics and Modeling in Mechanobiology, v. 10, p. 755-766. - S. J. Tan, Q. Li, C. T. Lim, and G. V. Shivashankar, 2010
Manipulation and isolation of single cells and nuclei
Nuclear Mechanics and Genome Regulation, v. 98, p. 79-96. - Q. Li, C. T. Lim, 2010
Structure mechanical property changes in nucleus arising from breast cancer
Cellular and Biomolecular Mechanics and Mechanobiology, v. 4, p. 465-475. - H. W. Hou, Q. Li, G. Y. H. Lee, A. P. Kumar, C. N. Ong, and C. T. Lim, 2009
Deformability study of breast cancer cells using microfluidics
Biomedical Microdevices, v. 11, p. 557-564. - Q. Li, G. Y. H. Lee, C. N. Ong, and C. T. Lim, 2008
AFM indentation study of breast cancer cells
Biochemical and Biophysical Research Communications, v. 374, p. 609-613. 1.
MechanoMedicine Instruments

Qingsen Li
Qingsen Li is a multidisciplinary scientist with expertise in mechanical engineering, bioengineering, and mechanobiology. Since 2018, he has led the Cancer Engineering R&D Core Facility at IFOM, where he develops cutting-edge biomedical technologies for cancer research and clinical applications. After earning his degree in Engineering Mechanics from Huazhong University of Science and Technology in 2003, Qingsen pursued his master’s research in computational simulation for bridge design. However, his growing interest in biology led him to the National University of Singapore, where he obtained a PhD in Bioengineering in 2010 under Prof. Chwee Teck Lim. His doctoral work provided groundbreaking insights into cancer cell mechanics, showing that breast cancer cells are softer than normal cells. He also developed microfluidic devices for high-throughput cancer cell detection. Qingsen then joined the Mechanobiology Institute in Singapore, where his research focused on how cells interact with their physical environment. Using advanced microfabrication techniques, he demonstrated that mechanical forces can regulate nuclear structure and function, unveiling a direct link between external physical cues and intracellular signaling. In 2015, he joined Marco Foiani’s lab at IFOM, where he pioneered the use of 3D printing and microfabrication to develop novel mechanobiology tools, including cell stretching and compression devices. Today, as head of the Cancer Engineering R&D Core Facility, he continues to drive innovation at the intersection of physics, engineering, and cancer research, developing technologies that enhance our understanding of tumor biology and its clinical implications.