Functional Genomics Screening

Unit info
CRISPR/Cas9 is a revolutionary gene-editing tool adapted from bacterial defense mechanisms. When infected, bacteria incorporate viral DNA segments into CRISPR (Clustered Regularly Interspaced Short Palindromic Repeats) sequences.
At the following viral attack, the Cas9 (CRISPR-associated protein 9) enzyme uses the RNA molecules transcribed from the CRISPR sequences to recognise the viral DNA and neutralize the threat.
In 2012, Nobel Prize winners Jennifer Doudna and Emmanuelle Charpentier adapted this system for use in eukaryotic cells, creating a precise genome-editing tool that cuts DNA and relies on cellular repair mechanisms to introduce genetic changes.
The Functional Genomics Screening (FGS) Unit specializes in high-throughput CRISPR screening using pooled libraries in diverse in vitro and in vivo models.
This technology assesses the effects of thousands of genetic perturbations at single-cell resolution with CRISPR knockout (CRISPR KO), CRISPR interference (CRISPRi), and CRISPR activation (CRISPRa).
It also utilizes base and prime editing to investigate specific mutations, such as those relevant to cancer development and progression.
The first approach combines a Cas module with a deaminase to convert DNA bases, while the second uses a template RNA to write the desired genetic changes into the target DNA. The FGS Unit integrates these perturbations with functional genomics readouts, including cellular viability and complex phenotypic analyses, often in collaboration with the Flow Cytometry Core Facility and the Experimental Therapeutics Program.
Comprehensive data analysis is supported by IFOM’s Research Computing & Data Science Unit. Collaborating with various IFOM groups, the FGS Unit aims to elucidate gene functions, understand their roles in disease mechanisms, and identify drug targets.
In addition to supporting IFOM researchers by employing, developing and optimizing CRISPR/Cas-based tools, the Unit adopts such technologies to investigate the DNA damage response at scale.
The Functional Genomics Screening Facility provides comprehensive, high-throughput CRISPR-based screenings tailored to cancer research, including CRISPR knockout (KO), interference (CRISPRi), activation (CRISPRa), and Base editing screens to systematically assess gene function in in vitro and in vivo models. In particular, the Facility offers:
- Custom assay development, including definition of the best experimental conditions for the screen
- Library design
- Library amplification, virus packaging and optimization of the transduction conditions
- Optimization of the screen functional readouts (in collaboration with the Flow Cytometry Facility for FACS-based screens)
- Screen execution, genomic DNA extraction, and NGS sample preparation
- Data analysis and interpretation in partnership with the IFOM’s Research Data & Biocomputing Science, ensuring rigorous data analysis and interpretation.
Collaboration
Collaborating PI in the framwork of Athena, IFOM’s strategic plan:
4D-Nucleofector® X Unit
Facility Acknowledgment
According to our Core Facilities Guidelines, if Facility services were used in work described in a publication, the Facility must be acknowledged, citing both the Facillity name and its Research Resource Identifier (RRID). A typically acknowledgement might look like:
The research described was supported by the IFOM Functional Genomic Screening Core Facility (RRID: SCR_026867)
Co‑Authorship
If a staff member of an IFOM or Cogentech Core Facility contributes substantially to a published work, they should be included as a co‑author.
More information is available in Recommended guidelines and Acknowledging and citing core facilities
Examples of substantial contributions that may justify co‑authorship include:
- Contribution to experimental design or optimization
- Development or significant adaptation of methods or protocols
- Advanced data analysis and interpretation
- Intellectual input influencing the scientific direction of the study
Routine technical support or standard service provision generally warrants acknowledgment rather than co‑authorship.
- Decitabine cytotoxicity is promoted by dCMP deaminase DCTD and mitigated by SUMO-dependent E3 ligase TOPORS CJ Carnie, MJ Gotz, CS Palma-Chaundler, P Weickert, A Wanders, ... The EMBO Journal, 1-27 (2024)
- Transcription-coupled repair of DNA–protein cross-links depends on CSA and CSB CJ Carnie, AC Acampora, AS Bader, C Erdenebat, S Zhao, E Bitensky, ... Nature Cell Biology, 1-14 (2024)
- Cockayne syndrome proteins CSA and CSB promote transcription-coupled repair of DNA-protein crosslinks independently of nucleotide excision repair C Carnie, A Acampora, A Bader, V Gupta, G D'Alessandro, ... Nature Research (2024)
- USP37 prevents premature disassembly of stressed replisomes by TRAIP OV Kochenova, G D'Alessandro, D Pilger, E Schmid, SL Richards, ... bioRxiv, 2024.09.03.611025 (2024)
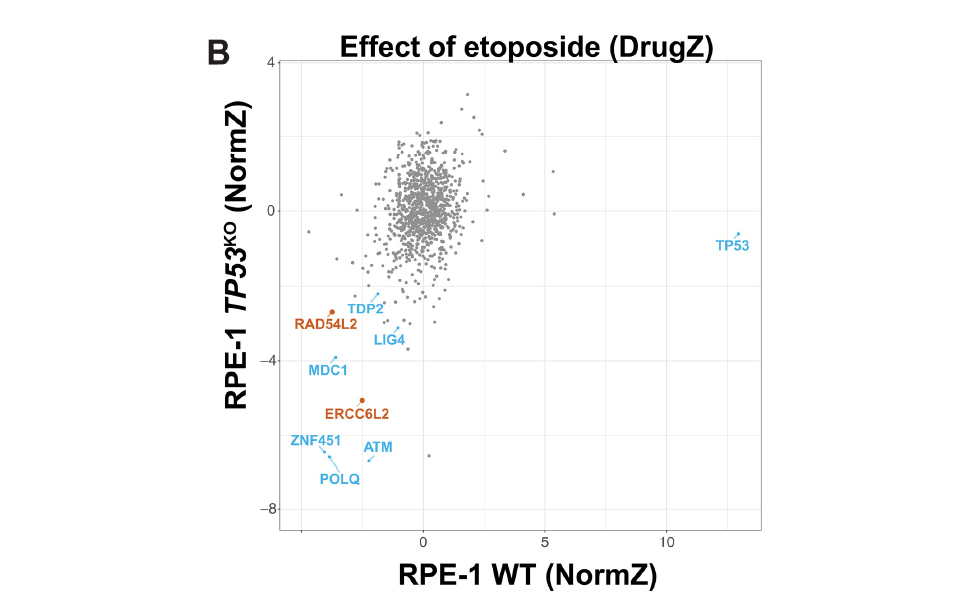
- RAD54L2 counters TOP2-DNA adducts to promote genome stability G D’Alessandro, DA Morales-Juarez, SL Richards, KC Nitiss, ... Science Advances 9 (49), eadl2108 (2023)
- The dCMP deaminase DCTD and the E3 ligase TOPORS are central mediators of decitabine cytotoxicity CJ Carnie, MJ Gotz, CS Palma-Chaundler, P Weickert, AR Wanders, ... bioRxiv, 2023.12.21.572728 (2023)
- BRCA2 controls DNA: RNA hybrid level at DSBs by mediating RNase H2 recruitment G D’Alessandro, DR Whelan, SM Howard, V Vitelli, X Renaudin, ... Nature Communications 9 (1), 5376 (2018)
- THE ROLE OF RNA AND DNA: RNA HYBRIDS AT DNA DOUBLE-STRAND BREAKS G D'Alessandro Università degli Studi di Milano (2018)
- Long non-coding RNA in the control of genome stability and cancer phenotypes G D’Alessandro, FA di Fagagna Non-coding RNA Investigation 2 (3) (2018)
- A role for RNA and DNA: RNA hybrids in the modulation of DNA repair by homologous recombination G D’Alessandro, M Adamowicz, D Whelan, SM Howard, ... bioRxiv, 255976 (2018)
- Transcription and DNA damage: holding hands or crossing swords? G D'Alessandro, FA di Fagagna Journal of Molecular Biology 429 (21), 3215-3229 (2017)
- A damaged genome’s transcriptional landscape through multilayered expression profiling around in situ-mapped DNA double-strand breaks F Iannelli, A Galbiati, I Capozzo, Q Nguyen, B Magnuson, F Michelini, ... Nature Communications 8 (1), 15656 (2017)
- Rearrangements of chromosome bands 15q12-q21 are secondary to HMGA2 deregulation in conventional lipoma G Macchia, KH Nord, G D'Alessandro, J Nilsson, L Magnusson, ... Oncology Reports 31 (2), 807-811 (2014)
- NEW FUSION GENE INVOLVING EWSR1 IN ACUTE MYELOID LEUKEMIA G Macchia, G D'Alessandro, C Lo Cunsolo, M Carella, O Palumbo, ... HAEMATOLOGICA 97 (s2), S133-S133 (2012)
Gallery

Giuseppina D’Alessandro
Giuseppina D’Alessandro holds a degree in Medical Biotechnology from the University of Bari and earned her PhD in Molecular Medicine from IFOM in Milan.
Her doctoral work, carried out in the laboratory of Dr. d’Adda di Fagagna, focused on the role of RNA and DNA:RNA hybrids in the repair of DNA double-strand breaks.
She then joined the University of Cambridge as a Marie Skłodowska-Curie iCARE-2 Fellow, working in the lab of Prof. Sir Steve Jackson.
During her postdoctoral training at the Gurdon Institute and the Cancer Research UK Cambridge Institute, she specialized in genome editing and DNA damage response, employing advanced CRISPR technologies including knock-out, inhibition, and base editing approaches.
After five years in the UK, Giuseppina returned to Italy to establish and lead the Functional Genomics Screening Core Facility at IFOM.
In this role, she applies her expertise in genome editing and screening to support IFOM researchers and explore DNA repair mechanisms at scale.
Giuseppina has led multiple research projects and coordinated international collaborations. She serves also a reviewer for international journals, including Nature Communications, and contributes to the preprint highlights service PreLights.